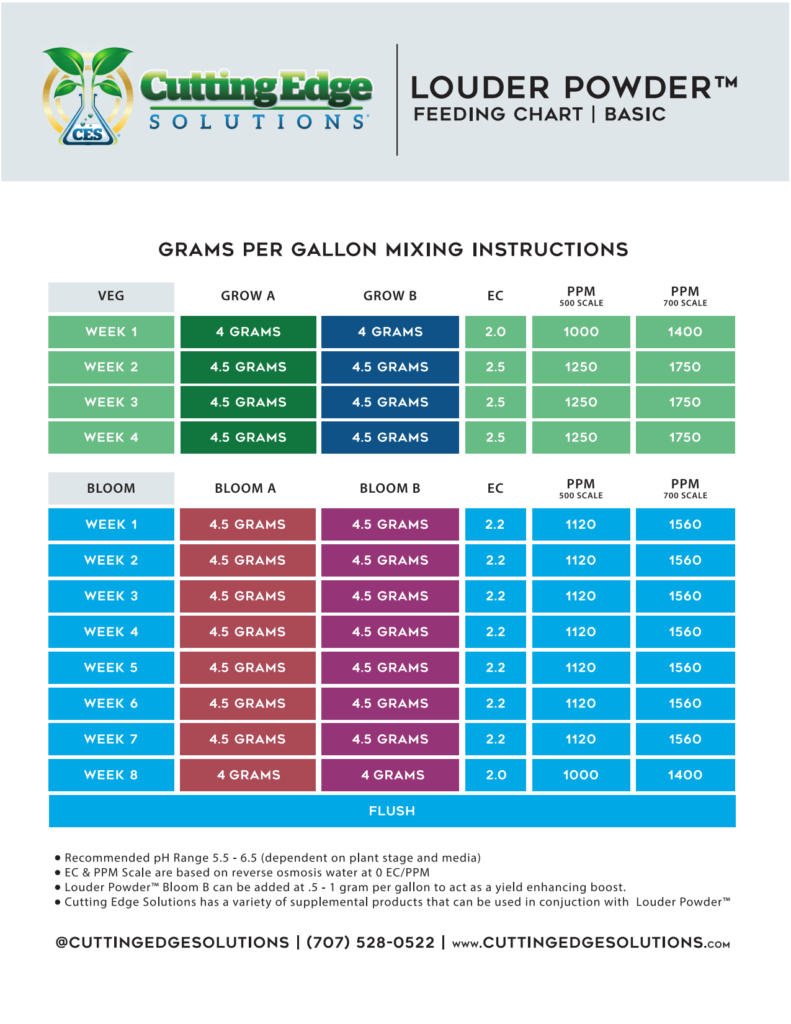
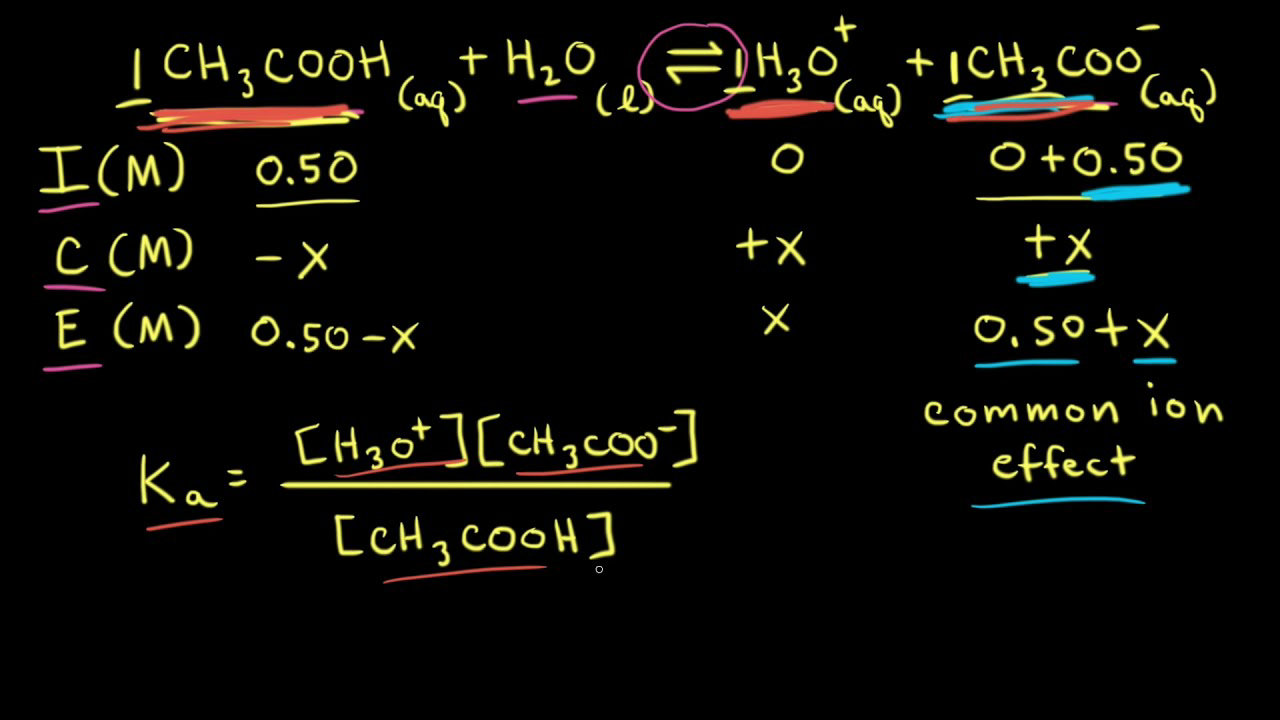Calculate the pH of a solution formed by mixing equal volumes of two solutions A and B of a strong acid having pH = 6 and pH = 4 respectively.
What is the pH of the resulting solution when the equal volume of 0.1M Noah and 0.01M HCL are mixed? - Quora

equilibrium - Calculation of the pH of a mixture of a strong acid and weak acid - Chemistry Stack Exchange